F gas – What is it and what you need to know?
Let’s talk about the duo of the hour – F gas and Ozone Depleting Substances (ODS) that are out to wreak havoc on the environment in their own horrific ways. While the distastefully named F-gas mounts up the temperature globally, the malicious ODS tears a hole in the upper of the ozone layer of the stratosphere. Surprisingly enough, F-Gases actually came up as a substitute to ODS after the Montreal Protocol since they did not pose any direct threat to the ozone, however, its major downside is that they’re powerful greenhouse gases.
Here, we delve deeper into understanding what the two substances are and the threats they pose to our environment and future.
Table of Contents:
- What are Ozone Depleting Substances (ODS)?
- What is F-Gas?
- What are the different F gases?
- The F gas Regulation
- List of F Gases
- Where are F-Gases used?
- Disadvantages of F gases?
- Reduction and Replacement
What are Ozone Depleting Substances (ODS)?
Ozone Depleting Substances (ODS) are a family of man-made chemicals that can cause damage to the earth’s atmosphere, particularly the stratospheric ozone layer. The three types of ODSs are:
- Chlorofluorocarbons (CFC)
- Bromofluorocarbons or Halons
- Hydrochlorofluorocarbons (HCFCs).
CFCs were largely used as refrigerants, aerosol propellants and solvents and are now completely phased out. Halons were used in fire protection applications and have also been completely phased out. HCFCs, however, represent the largest remaining use of ODS, which are mostly banned in the UK.
Other ODSs include Methyl Chloroform, Carbon Tetrachloride (the main precursor of CFCs) and Methyl Bromide.
In 1974, scientists Sherwood Rowland and Mario J. Molina discovered that chlorofluorocarbons (CFCs) could deplete Earth’s atmospheric ozone layer, which is essential in blocking the sun’s damaging ultraviolet rays. At the time, CFCs were in wide use in refrigerants and as propellants in aerosol sprays. Their inert and essentially nontoxic, characteristics made them well-suited for these applications but on the downside made them extremely harmful to the Earth. Although CFCs are inert in the lower troposphere, Rowland found that they can be broken down by UV radiation once they reach the stratosphere.

Source: Visual Capitalist
Finally, when the Antarctic ozone hole was found in 1985, the depletion of the ozone layer became a real and imminent danger. Lawmakers and the public at large took notice of the situation. And so 56 countries convened at the Montreal Protocol in 1987 to cut CFC production and use in half. Montreal Protocol became a global agreement to protect the stratospheric ozone layer by gradually phasing out the production and consumption of ODS. This landmark agreement successfully reduced the global production, consumption, and emissions of ODS, but there is still a long way to go.
What is F-Gas?
F Gas stands for fluorinated gas, the term is used to describe a particular family of man-made gases which are widely used in industries as well as certain home appliances. The F-gases are most commonly used as refrigerants in commercial refrigeration and air conditioning. We aren’t absolute strangers to the pesky F Gases, in fact, many of our essential appliances have them.
What are the different F gases?
F-gas stands for “fluorinated gas”. These are gases that contain fluorine atoms and are often used in industrial applications due to their unique properties such as their non-toxicity, non-flammability, and chemical stability.
However, F-gases are also potent greenhouse gases that contribute to climate change.
These F gas largely falls into 3 groups, hydrofluorocarbons (HFCs), perfluorocarbons (PFCs) & Sulphur hexafluoride (SF6). They were introduced as a replacement for ozone-depleting substances (ODS) like CFCs or Chlorofluorocarbon – which are being phased out as part of the Montreal Protocol. F-gas refrigerants are sometimes referred to by their ‘R-numbers’
HFC
Heavy application in industry offices, retail and much more. HFC is used as refrigerants in commercial fridges and air conditioning. Other uses include aerosols, solvent cleaning insulating foam, and fire protection.
PFCs
PFC has a few specialised applications and is used in semiconductor manufacturing as well.
USF6
Among a few specialised processes, USF6 is used in magnesium smelting and high-voltage switchgear.
Despite their drastic impact on the climate, fluorinated greenhouse gases (F gases) are used across many sectors of the UK economy as refrigerants, solvents, insulating gases, aerosols, or blowing agents, and can also be emitted as fugitive emissions from other manufacturing processes. The problem with F-gas lies in the fact that they are very powerful greenhouse gases. They trap heat and as a result, add to the rising global warming. F gases have a global warming potential (GWP) of up to 23,000 times that of CO2 (SF6), so the emission of even a small quantity of F gas to the atmosphere is harmful to the environment.
F-gas emissions accounted for 3% of UK greenhouse gas emissions in 2018. Emissions in 2018 were 37% below 1997, the year of highest emissions, as reduction technologies at halocarbon production plants have cut F-gas leakage by over 99%.
The F gas Regulation
What is the f gas regulation?
The F-Gas Regulation is a European Union law that aims to reduce emissions of fluorinated greenhouse gases (F-gases), which are potent greenhouse gases that contribute to climate change. The regulation imposes strict rules on the use, handling, and disposal of F-gases, with the goal of reducing their emissions by two-thirds by 2030 compared to 2014 levels. It also bans the use of certain F-gases in new equipment and sets limits on the number of F-gases that can be sold in the EU.
The regulation requires regular checks and maintenance of equipment containing F-gases. It also requires training and certification for personnel who handle these gases. It promotes the use of alternative technologies and substances with lower global warming potential. The regulation has been in effect since 2015 and is enforced by the European Chemicals Agency (ECHA) and national authorities in EU member states.
___
The aim of F-gas regulations?
- To reduce emissions of fluorinated greenhouse gases (F-gases), which are potent greenhouse gases that contribute to climate change.
- To reduce F-gas emissions by two-thirds by 2030 compared to 2014 levels.
- To promote the use of alternative technologies and substances with lower global warming potential.
- To set strict rules on the use, handling, and disposal of F-gases.
- To require regular checks and maintenance of equipment containing F-gases.
- To require training and certification for personnel who handle F-gases.
- To help mitigate climate change and reduce the impact of F-gases on the environment.
List of F Gases
The following is a list of common fluorinated gases that are regulated under the F-Gas Regulation:
Hydrofluorocarbons (HFCs)
Perfluorocarbons (PFCs)
Sulfur hexafluoride (SF6)
Nitrogen trifluoride (NF3)
Chlorofluorocarbons (CFCs)
Hydrochlorofluorocarbons (HCFCs)
They are commonly used in refrigeration and air conditioning systems, fire protection systems, electrical equipment, and other industrial applications.
Where are F-Gases used?
F-gases (fluorinated gases) are used in a wide range of applications including:
- Refrigeration and air conditioning: F-gases are used as refrigerants in air conditioning and refrigeration systems.
- Foam blowing agents: F-gases are used as blowing agents in the production of foam insulation materials such as rigid polyurethane foam.
- Fire protection: F-gases are used in fire extinguishing systems and as fire retardants in plastics.
- Electrical equipment: F-gases are used in electrical equipment such as transformers and switchgear.
- Aerosols: F-gases are used as propellants in aerosol sprays.
- Solvents: Some F-gases are used as solvents in various industrial applications.
Disadvantages of F gases?
F-gases (fluorinated gases) have several disadvantages, including:
- High Global Warming Potential (GWP): F-gases are potent greenhouse gases that have a high GWP compared to other greenhouse gases such as carbon dioxide. This means they have a much greater impact on climate change per unit of mass.
- Long atmospheric lifetimes: F-gases can remain in the atmosphere for many years, which contributes to their long-term impact on climate change.
- Ozone depletion: Some types of F-gases, such as chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs), can also contribute to ozone depletion in the stratosphere.
- Health and safety risks: Some F-gases, such as sulfur hexafluoride (SF6), are also classified as hazardous substances due to their potential health and safety risks to humans and the environment.
Reduction and Replacement
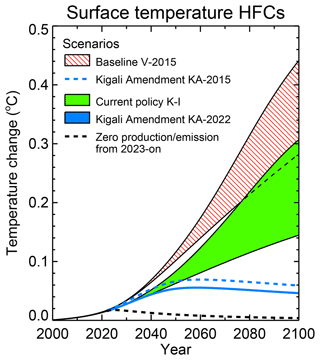
Reduction of emissions requires a targeted replacement of F gases by environmentally friendly alternatives or alternative technologies. There is an urgency to come up with mitigation policies and the need is getting acknowledged internationally. Kigali Amendment of 2016, the amendment to the Montreal Protocol, recognised this and its adoption set out a set of legally binding obligations for developing and industrial nations. It aims at phasing out F gases entirely over the course of the future. Due to their highly damaging impact on the climate, F-gases should be restricted to very limited uses where there are no viable alternatives.
The ABCs of Sustainability Development
There is a growing need to adopt a new set of Sustainable Development Goals that aim to “Transform the World”. Change comes with collective effort, mutual understanding and knowledge toward specific goals.
Despite the UN and other international organisations making crucial decisions, the result is far from expected. It made me wonder what it is that we are falling short on? Is it the lack of interest or lack of awareness?
That’s when I had a eureka moment, and it suddenly started to make all sense. Many people don’t know the complicated terms and references that are used when we talk about sustainability. The idea inspired me to create a new series called “The ABCs of Sustainability Development”. I hope that this series of blogs is well received and serves its purpose.
FAQs
What is F gas?
F gas stands for “fluorinated gas,” which are gases containing fluorine atoms that are commonly used in industrial applications.
·
What is F gas regulation?
The F Gas Regulation is a regulatory framework in the European Union aimed at reducing emissions of F-gases, which are potent greenhouse gases, and promoting the use of alternatives with lower global warming potential.
·
What is ODS (Ozone Depleting Substances)?
ODS stands for “Ozone Depleting Substances,” which are chemicals that can deplete the Earth’s ozone layer, resulting in increased UV radiation and potential harm to living organisms.
·
What is F-gas used for?
F-gases are commonly used in refrigeration and air conditioning systems, foam insulation materials, fire protection systems, electrical equipment, aerosol sprays, and solvents.
·
What are examples of F-gases?
Common examples of F-gases include hydrofluorocarbons (HFCs), perfluorocarbons (PFCs), sulfur hexafluoride (SF6), nitrogen trifluoride (NF3), chlorofluorocarbons (CFCs), and hydrochlorofluorocarbons (HCFCs).
·
Is F gas harmful to humans?
Some F-gases, such as sulfur hexafluoride (SF6), are classified as hazardous substances and can pose potential health and safety risks to humans and the environment. However, F-gases are generally considered safe when handled and used properly.
·
What are the disadvantages of F-gases?
F-gases have several disadvantages, including their high global warming potential, long atmospheric lifetimes, potential contribution to ozone depletion, and potential health and safety risks to humans and the environment. These disadvantages have led to increased efforts to limit their use and emissions, as well as to find alternatives with lower global warming potential and environmental impact. The F-Gas Regulation is an example of such efforts.





